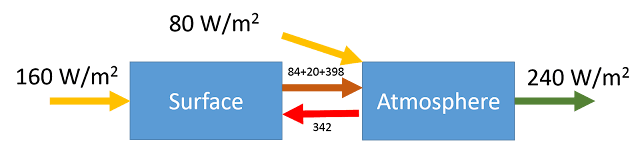
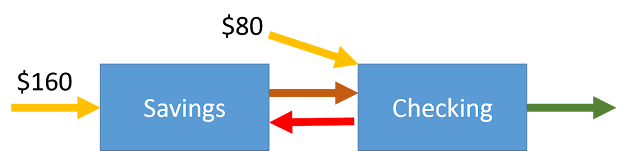
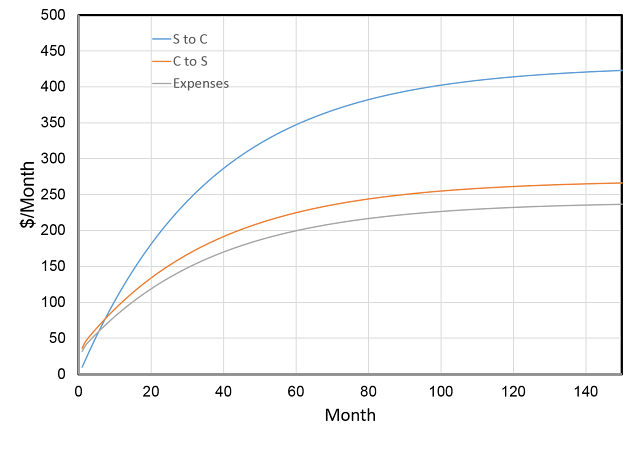
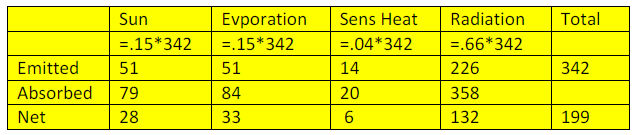
Reto Knutti wrote his experiences of Fake News and how to deal with it for the Schweiz am Wochenende. It also appeared on the ETH Zurich website. Since it is in German, Eli thought a translation would be useful. As a scientist he included a few footnotes.
------------------------------------------------------------------------------------------------------------------
Research in the Era of Fake News
Fake news is everywhere, but climate research is specially targeted. Reto Knutti on his experience with fake news and slander
Reto Knutti
A Russian website cites me under a picture as saying that mankind has only three quiet years left. As a climate scientist I supposedly had written a report to this effect that was under lock and key [1]. Or: I was supposed to have said in another report that mushroom spores cause hurricanes, earthquakes and tsunamis [2]. Of course this is all completely made up from the first to the last word. Still it did not take long for a Russian television station to come calling for an interview.
Communicating climate change is not for the faint of heart: The range of reactions to my public statements ranges from allegations of fraud, power seeking and greed to hand written “proof”, that claim to show that conservation of energy is really something else than you find in physics books. I’ve gotten used to this but it’s different when it comes to slander.

Those who lie about climate change or spread false rumors mostly do that because proposed solutions collide with their personal world view (Picture: marchmelena29 /iStock)
Known Symptons
The problem in a post factual age are at least superficially well known: Democracy requires an informed public that in spite of differing opinions find common solutions. But the opening of academic discourse (wissen-ER) through social media has led to each and every being able to say something faster and faster. Everybody talks, no one listens and experts are often held to be suspicious rather than trustworthy.
In social media we often find ourselves in bubbles, there are no quality controls and fantasies or controversial statements get the most “likes”. Fake news spread on Twitter faster and wider than facts [3].
There are reasons why the truth does not penetrate. Sometimes it’s just about paying attention or advertising revenue. In the case of climate there are often political or economic interests. In 1998 an internal report from Shell documented the dangers of manmade climate change and the possible effects on the oil industry. After that for many years the company tried to cast doubt on the scientific consensus [4].
Even today in the US one in four registered voters believes that global warming is fictional [5]. More than anywhere else opinions about climate change are determined by political ideology: People don’t “believe“ in climate change because the possible solutions (high energy prices, governmental regulations) contradict their personal neoliberal convictions about unlimited growth and small government.
Hand Wringing About Solutions
Much of this diagnosis is not new. The underlying problems are made worse by the vanishing of quality journalism, shouting on social media and as a result an increasingly polarized society. How can one best deal with Fake News?
I have no conclusive answer. Some answers which at least at first view seemed clear (not only to me) simply don’t work. More facts in even more reports are to be sure relevant for policy decisions and technical solution but they scarcely change the opinions of the already convinced. On the contrary, making clear why Fake News is fake often only increases its visibility.
Most of my attempts to respond to hostile or random claims and discuss them on line have not been fruitful. The exchanges give the impression that the situation is unclear and everything is open to debate. It is astounding that even penetrating the bubble does not help. People who every day voluntarily confront different perspectives on Twitter become even more convinced in their views [6].
Get Involved Anyhow
There are also bright spots. New work shows that readers can better deal with fake news if they are warned beforehand that it exists on a topic [7]. I am still convinced that we must continue to think about the relevant questions and discuss them in public despite the abundance of evidence and the shortage of time.
My experience is that eye to eye dialog is best when we are trying to separate facts from opinion. A thermometer is not politically right or left exactly the same as there are not two sides to gravity. We can agree about the facts and still have a debate about how we should react to them. As scientist I don’t dictate to society what should be done. However, I consider it my duty not only to produce numbers but to look at them critically and to explain what they mean without engaging in PR – a tightrope dance in the age when researchers are fighting for money and positions [8].
In addition to a common (fact based) denominator I always try to find common values and goals in conversations. For this the way a problem is formulated, so called framing, is extremely important. Respect for other opinions and readiness to listen are elements that build the trust that helps build bridges. In this stories are crucial to deliver the message. It all takes time but for me there is no way around it.
Sometimes It Needs a Few More Characters
Controversy delivers good headlines buy hardly constructive discussions. With all the enthusiasm for new media and big data, for me, a real discussion requires both the solid synthesis of a quality newspaper and an informed reader rather than propaganda tweets from Trolls on the Web. Not to mention algorithms that daily determine what is true or false. Jack Dorsey, co-founder of Twitter, once said: “One can change the world with 140 characters” He is really right. But to understand the world and to improve it for the next generation you need a few more.
References
1.
The ETH deliberately is not linking to these pages. A Google search “knutti "the impending weather and climate catastrophe" will find multiple examples
2.
The ETH deliberately is not linking to these pages. A Google search “knutti Monica Gagliano" will find it
3.
Article in Science
4.
Center for international and environmental law: Internal Documents Shed New Light on Shell’s Role in the Climate Crisis (April 2018)
5.
Yale Program on Climate Change Communication: Politics & Global Warming (March 2018)
6.
Article in the Tagesanzeiger based on a preprint of a scientific manuscript
7.
PNAS: Science in the age of selfies