Methane and Ozone
Some time ago, Eli discussed the atmospheric oxidation of methane a topic to which he now returns in the context of ozone pollution and the effects on air quality and human health. Tropospheric ozone is a result of NOx chemistry, where NOx, either referred to as NO ex, or nocks, is the sum of NO (nitric oxide) and NO2 (nitrogen dioxide) in the atmosphere.
Tropospheric ozone is formed by the photodissociation of NO2 between 400 and 300 nm, with the long wavelength limit set by the bond strength of O - NO, and the lower one by the absorption cut off of stratospheric ozone which absorbs all of the solar light below 300 nm (to be fussy 306 nm or so and as long as the bunnies are being fussy somewhere like 420 nm on the high end because of thermal population of excited vibrational levels)
NO2 + hν --> NO + O(3P) [1]
If Reaction 2 is assumed to be fast (it is because there is so much oxygen and nitrogen around) the rate at which ozone is produced is j [NO2] where j is the flux of UV light and the rate at which it is consumed is k3[NO][O3] where k3 is the rate constant for reaction 3[X] means the concentration of X. Under steady state conditions when production via Reaction 1 equals destruction via Reaction 3
which can be solved for an estimate of the ozone concentration
in terms of the steady state concentrations of NO and NO2
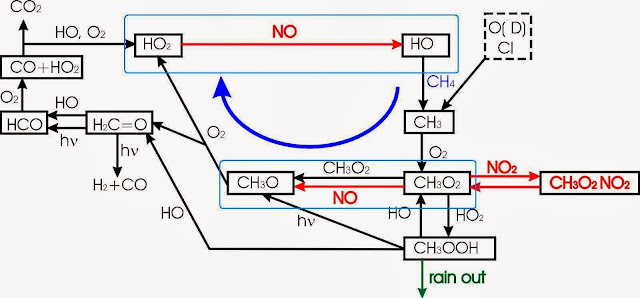
While reaction [3] appears to limit the amount of ozone that can form, other reactions which cycle NO back to NO2 without consuming ozone would lead to much higher ozone concentrations. Volatile organic compounds (VOCs) provide such a path, and methane is the VOC with the highest atmospheric concentration. Moreover, as a non-condensible gas not very susceptible to rain out, it spreads worldwide in ways that heavier VOCs do not. Looking at the reaction scheme for methane oxidation






7 comments:
"CO2 and formic acid (in droplets) is produced by sunlight when methane (and other VOCs) escape to air. With aldehydes and alcohols the reaction is a bit different and produces less H+ than with plain hydrocarbons. We won't go into more complex compounds here, and you don't need to memorize the following reaction pathway, but there it is to prove the connection between tropospheric ozone, VOCs and (formic) acidic rain. The important thing is to remember is that in atmospheric chemistry many of the compounds are in excess and once the activation energy of these is reached all sorts of reactions happen.", crash course on environmental chemistry (pretty excactly 15 years ago). (some speculation about the chemistry in Northern Lights afterwards)
Thanks for the memories and reminding there's so much stuff happening about that I'm not going to learn lest fully understand. :-)
I love it when you talk dirty Eli.
Bernard J.
So to ask either the obvious or obviously wrong, is the global anthropogenic increase in methane levels causing significant, direct harm to human health?
(I gave your Keeling people $50, since it seemed to be odd to be supporting it with words only).
This must mean there are higher levels of tropospheric ozone around gas mining sites right? I imagine this is fairly common knowledge but I haven't heard anything about increased ozone around areas where fracking is prevalent. The only time I hear about it is on hot days around the city.
You need UV light, NOx AND VOCs. Mines tend to have large diesel engines which are sources of NOx, so, yep this is something you might expect, at least for open pit.
" is the global anthropogenic increase in methane levels causing significant, direct harm to human health?"
Yes. As an example, see work by Jason West at UNC.
-MMM
Post a Comment